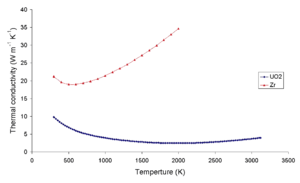
Uranium dioxide
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Read other articles:

1960 film by Charles Vidor, George Cukor Song Without EndVHS coverDirected byCharles VidorGeorge CukorWritten byOscar MillardProduced byWilliam GoetzStarringDirk BogardeCapucineGeneviève PageCinematographyJames Wong HoweEdited byWilliam LyonMusic byMorris StoloffHarry SukmanFranz LisztDistributed byColumbia PicturesRelease date August 11, 1960 (1960-08-11) Running time141 min.CountryUnited StatesLanguageEnglishBudget$3.5 million[1]Box office$1,500,000 (US and Canada re...

У этого термина существуют и другие значения, см. Башкирский. Башкирский язык Самоназвание башҡорт теле,башҡортса Страны Россия, Украина[1], Казахстан[2], Узбекистан[3] Регионы Башкортостан, Челябинская область, Оренбургская область, Тюменская область, Свердлов

2019 book by Robert Caro For other uses, see Working. Working: Researching, Interviewing, Writing First edition (US)AuthorRobert CaroSubjectMemoirPublisherKnopf (US)Bodley Head (UK)Publication dateApril 9, 2019 (2019-04-09)Pages240ISBN9780525656340 External videos C-SPAN interview with the author, 2019 Working: Researching, Interviewing, Writing is a memoir by biographer Robert Caro about the craft of biographical research and writing. Further reading Detrow, Scott (April 8, 20...

Stasiun Rangkasbitung R22LM01 Kondisi peron Stasiun Rangkasbitung.Nama lainStasiun RangkasLokasiJalan Stasiun Rangkasbitung No.1Muara Ciujung Timur, Rangkasbitung, Lebak, Banten 42314IndonesiaKetinggian+22 mOperatorKereta Api Indonesia Daerah Operasi I Jakarta KAI CommuterLetak dari pangkal km 79+694 lintas Angke–Tanah Abang–Rangkasbitung–Merak km 0+000 lintas Rangkasbitung–Labuan[1] Jumlah peron3 (satu peron sisi dan dua peron pulau bertangga tinggi)Jumlah jalur4 jalur 1: sep...

Ein FW3/22 Pillbox-Bunker nahe dem Kennet-und-Avon-Kanal Die General Headquarters Line (auch als GHQ Line bezeichnet) war eine britische, während des Zweiten Weltkrieges angelegte Verteidigungslinie. Diese als Hauptverteidigungsstellung angelegte Linie sollte den Großraum London und das mittelenglische Industriegebiet schützen. Dieses Verteidigungssystem beginnt am Nordende der Taunton Stop Line nahe dem Ort Highbridge in Somerset, verläuft dann längs des Flusses Brue und des Kennet-und-...

Wappen Deutschlandkarte 53.459779.4481119Koordinaten: 53° 28′ N, 9° 27′ O Basisdaten Bundesland: Niedersachsen Landkreis: Stade Samtgemeinde: Harsefeld Höhe: 19 m ü. NHN Fläche: 26,5 km2 Einwohner: 2126 (31. Dez. 2022)[1] Bevölkerungsdichte: 80 Einwohner je km2 Postleitzahl: 21698 Vorwahl: 04164 Kfz-Kennzeichen: STD Gemeindeschlüssel: 03 3 59 005 Gemeindegliederung: 2 Ortsteile Adresse der Gemeindeverwaltu...

سيرهي بويكو معلومات شخصية الميلاد 30 يونيو 1977 (46 سنة) بيلهورود دنيستروفسكيي الطول 1.82 م (5 قدم 11 1⁄2 بوصة) مركز اللعب حارس مرمى الجنسية أوكرانيا المسيرة الاحترافية1 سنوات فريق م. (هـ.) 1995–1996 [[FC DnistrovetsBilhorod-Dnistrovskyi|FC DnistrovetsBilhorod-Dnistrovskyi]] [لغات أخرى]...

ألكساندرة كورنهاوزر فريزر (بالسلوفينية: Aleksandra Kornhauser Frazer) معلومات شخصية اسم الولادة (بالسلوفينية: Aleksandra Caleari) الميلاد 26 سبتمبر 1926 شكوفجا لوكا الوفاة 17 مايو 2020 (93 سنة) [1] ليوبليانا مواطنة يوغوسلافيا (1926–1992) سلوفينيا (1992–2020) عضوة في الأكاديمية ا

لويس كوبيلا (بالإسبانية: Luis Cubilla) معلومات شخصية الميلاد 28 مارس 1940(1940-03-28)بايساندو الوفاة 3 مارس 2013 (عن عمر ناهز 72 عاماً)أسونسيون سبب الوفاة سرطان المعدة الطول 170 سنتيمتر مركز اللعب مهاجم الجنسية الأوروغواي مسيرة الشباب سنوات فريق Colón de Paysandú المسيرة...

Maria Rosaria (Mara) Carfagna (kelahiran 18 Desember 1975) adalah seorang politikus Italia Latar belakang Carfagna lahir di Salerno Pranala luar Ministry biography Diarsipkan 2008-05-12 di Wayback Machine. Personal blog Diarsipkan 2009-05-28 di Wayback Machine. Mara Carfagna - slideshow and video by The Huffington Post lbsKabinet Berlusconi IV (2008–11) Berlusconi Tremonti Maroni Alfano Palma Frattini Sacconi Fazio Gelmini La Russa Scajola Romani Zaia Romano Prestigiacomo Matteoli Bondi Gal...

Chinese philosophies flourishing in the 500s–221 BC Hundred Schools of ThoughtTraditional Chinese諸子百家Simplified Chinese诸子百家TranscriptionsStandard MandarinHanyu Pinyinzhūzǐ bǎijiāWade–Gileschu1-tzu3 pai3-chia1IPA[ʈʂútsɨ̀ pàɪtɕjá]WuRomanizationTsoe tzy ba' gaYue: CantoneseYale RomanizationJyū-jí baak-gāaJyutpingZyu1-zi2 baak3-gaa1Southern MinTâi-lôTsu-tsú pah-ka The Hundred Schools of Thought (Chinese: 諸子百家; pinyin&...

Overview of music traditions in Lithuania: folk, classical, contemporary, pop, etc. This article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these template messages) This article's lead section may be too short to adequately summarize the key points. Please consider expanding the lead to provide an accessible overview of all important aspects of the article. (March 2012) This article's use of external links may not follow...

لمعانٍ أخرى، طالع بشت (توضيح). بستمعلومات عامةالتقسيم الإداري مقاطعة بشت البلد المجر الإحداثيات 47°30′N 19°06′E / 47.5°N 19.1°E / 47.5; 19.1 تعديل - تعديل مصدري - تعديل ويكي بيانات القسم الشرقي من العاصمة بودابست (بشت) 47°30′N 19°6′E / 47.500°N 19.100°E / 47.500; 19.100 بس...

СокльєрSauclières Країна Франція Регіон Окситанія Департамент Аверон Округ Мійо Кантон Нант Код INSEE 12260 Поштові індекси 12230 Координати 43°58′37″ пн. ш. 3°22′04″ сх. д.H G O Висота 580 - 1 341 м.н.р.м. Площа 38,81 км² Населення 169 (01-2020[1]) Густота 4,12 ос./км² Розміщення Вл�...

School van de bevrijdingsbeweging PAIGC in de bevrijde gebieden, Guinee-Bissau, 1974. Albert Anker (1896) Een school is een instelling waarin onderwijs op een bepaald niveau gegeven wordt. Vanwege de leerplichtwet gaan de meeste kinderen naar een school, hoewel ook andere vormen van onderwijs mogelijk zijn om aan de leerplicht te voldoen. Het woord 'school' is afgeleid van het Griekse 'σχολή', dat 'vrije tijd' betekent. Onderwijs was namelijk oorspronkelijk iets waarvoor men vrije tijd m...

Line separating Armenian and Azerbaijani forces in the Nagorno-Karabakh conflict The Nagorno-Karabakh line of contact (1994–2020) in red, with the largely unguarded Murovdag (Mrav) mountain range in the north. The Line of Contact (Armenian: շփման գիծ, shp’man gits, Azerbaijani: təmas xətti) was the front line which separated Armenian forces (the Nagorno-Karabakh Defense Army and the Armenian Armed Forces) and the Azerbaijan Armed Forces from the end of the First Nagorno-Karabakh...

2014 Indian filmVegamDirected byK G Anil KumarWritten byK G Anil KumarScreenplay byK G Anil KumarStarringVineeth KumarJacob GregorySamskruthy ShenoyPratap PothenShammi ThilakanCinematographyManoj Kumar KhatoiEdited byDeepu S. JosephMusic byGovind MenonProductioncompanyFX4 Movie MakersRelease date 11 July 2014 (2014-07-11) (Kerala) CountryIndiaLanguageMalayalam Vegam is a 2014 Malayalam film starring Vineeth Kumar,[1] Jacob Gregory, Samskruthy Shenoy, Prathap Pothen,...

Frans Johan Louwrens GhijselsBiografiKelahiran8 September 1882 Kematian2 Maret 1947 (64 tahun)Data pribadiPendidikanUniversitas Teknik Delft KegiatanPekerjaanArsitek dan Perencana kota Bekerja diAlgemeen Ingenieurs- en Architectenbureau (AIA) (en) Frans Johan Louwrens Ghijsels (8 September 1882 – 2 Maret 1947) dulu adalah seorang arsitek dan perencana perkotaan yang bekerja di Belanda dan Hindia Belanda.[1] Ghijsels adalah pendiri AIA, konsultan arsitektur terbesar di ...

American street performer Naked CowboyBornRobert John Burck (1970-12-23) December 23, 1970 (age 52)Cincinnati, Ohio, U.S.Occupation(s)Singer, songwriter, writer, former political candidate, actorYears active1998–presentSpouse(s) Patricia Burck, The Naked Cowgirl (m. 2013)Websitenakedcowboy.com Robert John Burck (born December 23, 1970), better known as the Naked Cowboy, is an American street performer, singer, songwriter, and occasional actor...

2011 South Korean filmHanjiTheatrical release posterDirected byIm Kwon-taekWritten byIm Kwon-taek Song Gil-hanProduced byMin Byung-lock Lee Hee-wonStarringPark Joong-hoon Kang Soo-yeon Ye Ji-wonCinematographyKim Hoon-kwangEdited byPark Soon-dukMusic byKim Soo-chulProductioncompanyJeonju International Film FestivalRelease date March 17, 2011 (2011-03-17) Running time118 minutesCountrySouth KoreaLanguageKoreanBudgetUS$1.73 millionBox officeUS$310,073 Hanji (Korean: 달�...